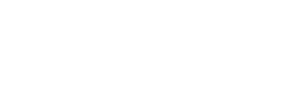Headline wins
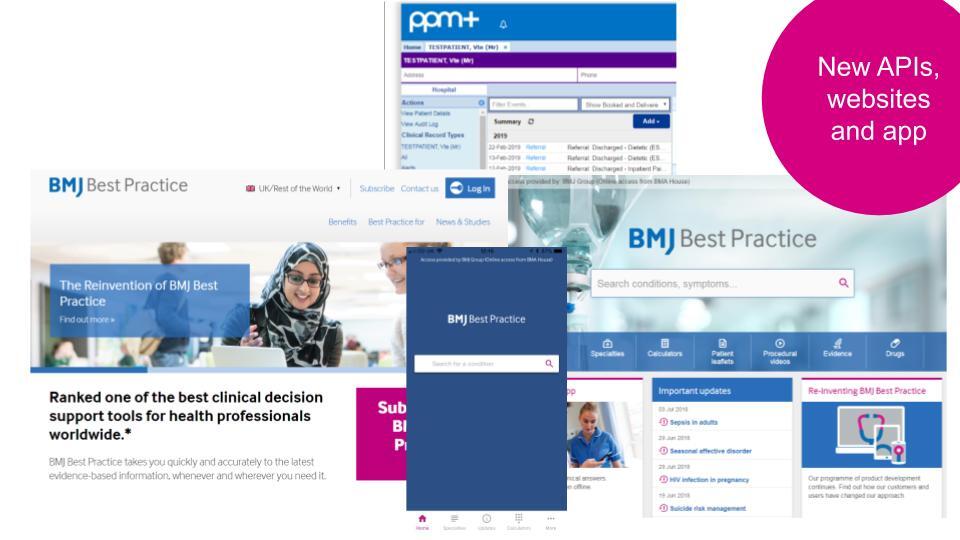
- The relaunch of BMJ Best Practice website and app, powered by new APIs, which saw a 202% growth in traffic and over 1 million users per month. Customer renewal rates reached 95%
- New business opportunities enabled by the flexible data structure, allowing BMJ to pursue new partnerships and integrations, including seamless EHR integration and international licensing. This led to winning important tenders and expanding the reach of BMJ Best Practice
- Streamlined content creation and management processes resulting in increased production capacity and a 40% reduction in production time
- Future product development, demonstrated by the launch of a new comorbidities tool. This tool leverages the granular content structure to address the complex needs of patients with multiple conditions
BMJ is a healthcare knowledge provider that advances healthcare worldwide by sharing knowledge and expertise to improve experiences, outcomes and value.
BMJ’s leading clinical decision support tool, BMJ Best Practice, is used by clinicians at the point of care in many countries. Offering advice on symptom evaluation, test ordering and treatment approaches for over 800 conditions in 30 specialties, BMJ Best Practice provides a rich source of expertise that healthcare professionals rely on every day.
Business challenge
BMJ wanted to innovate their product and user experience in line with clinicians’ needs and expectations for concise evidence-based information within the clinical setting. But the content was being created, stored and presented as long form articles typically extending to thousands of words. This made it difficult to improve the user interface and limited BMJ’s ability to offer content slices to third parties for integration into granular software systems, particularly EHR (Electronic Health Record) systems. The underlying editorial production system was reaching end of life and as such was slow to use and hard to maintain.
BMJ therefore took a strategic decision to build a completely new editorial production system, and crucially, to change the underlying data structure of the content to make it more granular. This would enable the company to satisfy current market demands, create new market opportunities and provide a springboard for further product innovation.
The solution
BMJ asked 67 Bricks to develop “Knowledge Base”, a bespoke editorial production system designed to be flexible enough meet BMJ’s needs, both now and in the future. 67 Bricks conducted a preparatory workshop with BMJ stakeholders to explore the ambition for the system and how best to work together to achieve it. The project was run on agile principles in two phases: elaboration and build. It was a big project with a limited timescale and so careful planning was essential. Key factors included:
- Strong prioritisation. This was critically important. BMJ adopted the ‘pragmatic marketing’ product development framework: this ensured that all the features built were based on external requirements.
- Prototyping. Shortlisted technologies including the data store and the XML editor were prototyped with real data so that stakeholders could see what it would mean for them in their daily activities.
- Co-development. BMJ’s and 67 Bricks’ developers worked together as one co-development team, but in different locations. This was facilitated through heavy use of online communication channels, in this case Slack and Trello, combined with daily joint stand-ups. There was reciprocal code review. The team were able to talk openly about how to implement features, and there was real trust. A shared understanding of the business drivers meant that everyone on the team was working towards a common goal. Co-development ensured maximum knowledge transfer to BMJ’s developers who now continue to maintain and extend the system to meet future needs.
- Content model. The new model, composed of individually referenceable fragment types (e.g. assessment, diagnosis), was refined iteratively throughout the project. More standardised than the old model, it provides the scope to slice up content, reuse content, work with partners, feed the product website and innovate.
- Meeting current needs. As well as providing for the future, it was essential to replicate current data outputs, to ensure no disruption in service to existing products and partners.
- Driving the BMJ Best Practice website. The development of Knowledge Base was undertaken in tandem with a separate project to redevelop the customer-facing product website. Therefore a Knowledge Base API was built to allow the new website to retrieve and use the content to power innovative front-end features.
- Content enrichment. Entity recognition was used to identify drug names and diagnoses within the editorial system. These enhanced terms are made available through the API and are retrieved by the new BMJ Best Practice website which can then use this information to deliver an enhanced search capability. The enriched terms also provide a basis for further innovation.
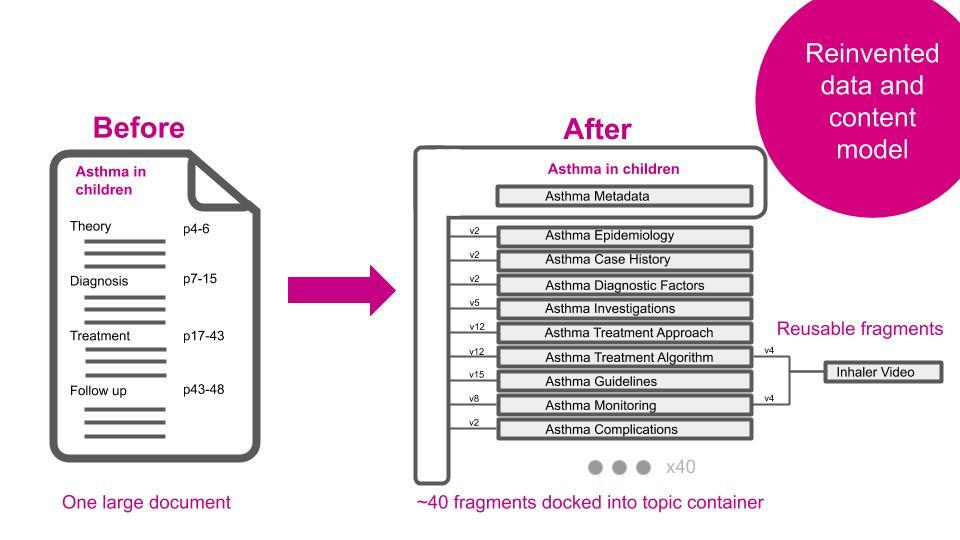
Using the new interface, editors are able to make changes to the relevant content fragments only and no longer need to read a long document in order to pinpoint the place(s) that needs updating.
Much of the content is available in several languages and so when a fragment is updated, only that fragment needs to be sent for re-translation, saving on time and expenditure.
Where needed, e.g. when an update is reviewed by an external expert, the system is able to reconstruct the long form version of the content so that the expert can see the change in context. A full audit trail is also available showing all changes and who made each one.
Business value
The engagement with 67 Bricks has enabled BMJ to reap benefits across the full content lifecycle from content creation to new product development. BMJ’s internal staff now have a much improved content production system which is responsive, collaborative, quicker to use and supported by a powerful faceted search function. It has enabled them to upskill their technology team, reduce operational costs and free themselves from technical debt and excessive manual work.
The APIs created were used to power a relaunched Best Practice website and app with innovative new product features such as alerts on the latest evidence changes and practical videos of common procedures. This led to a significantly better user experience, an impressive 202% growth in traffic with over 1 million users a month, and a 95% customer renewal rate. As a result of the product relaunch 96% of customers said that Best Practice helped them deliver improved patient care.
The new, flexible data structure opens up significant partnership opportunities and has enabled seamless EHR integration with multiple trusts. BMJ has been successful in winning several important tenders with Best Practice now available to all NHS health professionals in England, Scotland and Wales.
Crucially, BMJ have been able to springboard future product development and are now well placed to innovate. The granular, flexible data structure was instrumental in their ability to launch a new comorbidities tool in 2020, which is the first clinical decision support tool to improve care for patients with more than one condition. This unique tool will enable hospitals to improve patient outcomes, reduce costs, avoid unnecessary treatments and reduce length of stay.
As a heritage brand BMJ are proud to be able to deliver new products that are right at the forefront of evolving user needs. This has not gone unnoticed in the industry and BMJ Best Practice and 67 Bricks were delighted to be selected as finalists for the 2019 ALPSP Award for Innovation.
The BMJ Best Practice website is available at bestpractice.bmj.com